|
 Dmitri Ivanovich Mendeleev (1834-1907)
Dmitri Ivanovich Mendeleev (1834-1907)
 HO
ORGANIZED THE ALPHABET? We
will never be able to attribute to a single individual the development
of the basic building blocks of writing. Yet we do know the name of
the man who devised the method of classifying the basic building blocks
of matter. Dmitri Ivanovich Mendeleev was born in Siberia in 1834.
When Mendeleev became a professor of general chemistry at the University
of St. Petersburg, he was unable to find an appropriate textbook and
thus began writing his own. That textbook, written between 1868 and
1870, would provide a framework for modern chemical and physical theory. HO
ORGANIZED THE ALPHABET? We
will never be able to attribute to a single individual the development
of the basic building blocks of writing. Yet we do know the name of
the man who devised the method of classifying the basic building blocks
of matter. Dmitri Ivanovich Mendeleev was born in Siberia in 1834.
When Mendeleev became a professor of general chemistry at the University
of St. Petersburg, he was unable to find an appropriate textbook and
thus began writing his own. That textbook, written between 1868 and
1870, would provide a framework for modern chemical and physical theory.
|
 |
| Mendeleev first trained as a teacher in
the Pedagogic Institute of St. Petersburg before earning an
advanced degree in chemistry in 1856. |
|
 |
| Mendeleev’s first sketch of a periodic table of the elements |
|
|
 Elements and Their Properties
Elements and Their Properties
 OMBINATIONS
OF 26 LETTERS make
up every word in the English language. Similarly, all material things
in the world are composed of different combinations of about 100
different elements. An element is a substance that cannot be broken
down into simpler substances through ordinary chemistry--it is not
destroyed by acids, for example, nor changed by electricity, light,
or heat. Although philosophers in the ancient world had a rudimentary
concept of elements, they were incorrect in identifying water, for
example, as one. Today it is common knowledge that water is a compound,
whose smallest unit is a molecule. Passing electricity through a
molecule of water can separate it into two atoms of hydrogen and
one atom of oxygen, each a separate element. OMBINATIONS
OF 26 LETTERS make
up every word in the English language. Similarly, all material things
in the world are composed of different combinations of about 100
different elements. An element is a substance that cannot be broken
down into simpler substances through ordinary chemistry--it is not
destroyed by acids, for example, nor changed by electricity, light,
or heat. Although philosophers in the ancient world had a rudimentary
concept of elements, they were incorrect in identifying water, for
example, as one. Today it is common knowledge that water is a compound,
whose smallest unit is a molecule. Passing electricity through a
molecule of water can separate it into two atoms of hydrogen and
one atom of oxygen, each a separate element.
The ancient concept
of elements jibed with today's in noting that elements had characteristic
properties. Just as people not only look different from each other
but also interact differently with others, so elements have both
physical and chemical properties. Some elements form shiny solids,
for example, that react readily and sometimes violently with oxygen
and water. The atoms of other elements form gases that scarcely
interact with other elements.
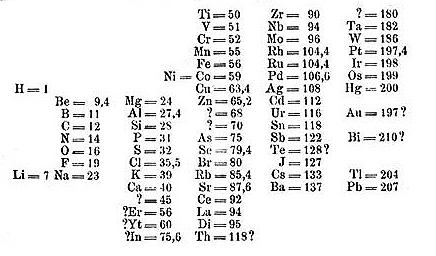 Mendeleev’s table as published in 1869, with many gaps and uncertainties Mendeleev’s table as published in 1869, with many gaps and uncertainties |
Below: The table, rotated ninety degrees, as shown in textbooks in 1898 when Marie Curie discovered Radium. Mendeleev and others promptly added the new element to their textbooks. Its place is in column II below Ba, Barium. (From W. Ostwald, Grundriss der Allgemeine Chemie.)
|
 CIENTISTS
HAD IDENTIFIED over 60 elements by Mendeleev's time. (Today
over 110 elements are known.) In Mendeleev's day the atom was considered
the most basic particle of matter. The building blocks of atoms
(electrons, protons, and neutrons) were discovered only later. What
Mendeleev and chemists of his time could determine, however, was
the atomic weight of each element: how heavy its atoms were in comparison
to an atom of hydrogen, the lightest element. CIENTISTS
HAD IDENTIFIED over 60 elements by Mendeleev's time. (Today
over 110 elements are known.) In Mendeleev's day the atom was considered
the most basic particle of matter. The building blocks of atoms
(electrons, protons, and neutrons) were discovered only later. What
Mendeleev and chemists of his time could determine, however, was
the atomic weight of each element: how heavy its atoms were in comparison
to an atom of hydrogen, the lightest element.
“I
began to look about and write down the elements with their
atomic weights and typical properties, analogous elements
and like atomic weights on separate cards, and this soon
convinced me that the properties of elements are in periodic
dependence upon their atomic weights.”
--Mendeleev, Principles of Chemistry, 1905, Vol. II
|
|
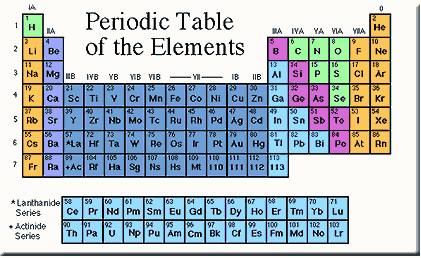
A modern periodic table.
|
 Classifying
the Elements
Classifying
the Elements
 N
OVERALL UNDERSTANDING of how the elements are related
to each other and why they exhibit their
particular chemical and physical properties was slow in coming.
Between 1868 and 1870, in the process of writing his book, The
Principles of Chemistry, Mendeleev created a table or chart
that listed the known elements according to increasing order of
atomic weights. When he organized the table into horizontal rows,
a pattern became apparent--but only if he left blanks in the table.
If he did so, elements with similar chemical properties appeared
at regular intervals--periodically--in vertical columns on the table. N
OVERALL UNDERSTANDING of how the elements are related
to each other and why they exhibit their
particular chemical and physical properties was slow in coming.
Between 1868 and 1870, in the process of writing his book, The
Principles of Chemistry, Mendeleev created a table or chart
that listed the known elements according to increasing order of
atomic weights. When he organized the table into horizontal rows,
a pattern became apparent--but only if he left blanks in the table.
If he did so, elements with similar chemical properties appeared
at regular intervals--periodically--in vertical columns on the table.
Mendeleev was bold
enough to suggest that new elements not yet discovered would be
found to fill the blank places. He even went so far as to predict
the properties of the missing elements. Although many scientists
greeted Mendeleev's first table with skepticism, its predictive
value soon became clear. The discovery of gallium in 1875, of scandium
in 1879, and of germanium in 1886 supported the idea underlying
Mendeleev's table. Each of the new elements displayed properties
that accorded with those Mendeleev had predicted, based on his realization
that elements in the same column have similar chemical properties.
The three new elements were respectively discovered by a French,
a Scandinavian, and a German scientist, each of whom named the element
in honor of his country or region. (Gallia is Latin for France.)
Discovery of a new element had become a matter of national pride--the
rare kind of science that people could read about in newspapers,
and that even politicians would mention.
Claiming a new element
now meant not only identifying its unique chemical properties, but
finding the atom's atomic weight so the element could be fitted
into the right slot in the periodic table. For radioactive atoms
that was a tough challenge. At first these atoms were isolated only
in microscopic quantities. The straightforward way to identify them
was not by their chemical properties at all, but by their radiations.
Until the radioactive atoms could be sorted out with traditional
chemistry, some scientists were reluctant to call them new elements.
 HAT
MADE THE TABLE PERIODIC?
The value of the table gradually became clear, but not its meaning.
Scientists soon recognized that the table's arrangement of elements
in order of atomic weight was problematic. The atomic weight of
the gas argon, which does not react readily with other elements,
would place it in the same group as the chemically very active solids
lithium and sodium. In 1913 British physicist Henry Moseley confirmed
earlier suggestions that an element's chemical properties are only
roughly related to its atomic weight (now known to be roughly equal
to the number of protons plus neutrons in the nucleus). What really
matters is the element's atomic number—the number of protons
its atom carries, which Moseley could determine with X-rays. Ever
since, elements have been arranged on the periodic table according
to their atomic numbers. The structure of the table reflects the
particular arrangement of the electrons in each type of atom. Only
with the development of quantum mechanics in the 1920s did scientists
work out how the electrons arrange themselves to give the element
its properties. HAT
MADE THE TABLE PERIODIC?
The value of the table gradually became clear, but not its meaning.
Scientists soon recognized that the table's arrangement of elements
in order of atomic weight was problematic. The atomic weight of
the gas argon, which does not react readily with other elements,
would place it in the same group as the chemically very active solids
lithium and sodium. In 1913 British physicist Henry Moseley confirmed
earlier suggestions that an element's chemical properties are only
roughly related to its atomic weight (now known to be roughly equal
to the number of protons plus neutrons in the nucleus). What really
matters is the element's atomic number—the number of protons
its atom carries, which Moseley could determine with X-rays. Ever
since, elements have been arranged on the periodic table according
to their atomic numbers. The structure of the table reflects the
particular arrangement of the electrons in each type of atom. Only
with the development of quantum mechanics in the 1920s did scientists
work out how the electrons arrange themselves to give the element
its properties.
[On
learning about the table] “For the first time I saw
a medley of haphazard facts fall into line and order. All
the jumbles and recipes and hotchpotch of the inorganic
chemistry of my boyhood seemed to fit themselves into the
scheme before my eyes — as though one were standing
beside a jungle and it suddenly transformed itself into
a Dutch garden.”
— C.P. Snow
|
� 2000 -
American Institute of Physics
|
|



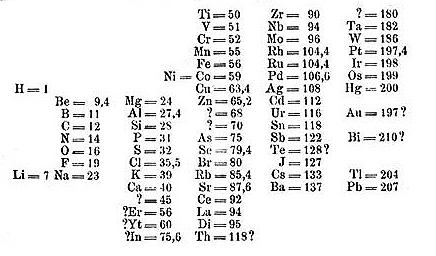 Mendeleev’s table as published in 1869, with many gaps and uncertainties
Mendeleev’s table as published in 1869, with many gaps and uncertainties